Colligative Properties - Boiling Point Elevation, Freezing Point Depression & Osmotic Pressure - YouTube
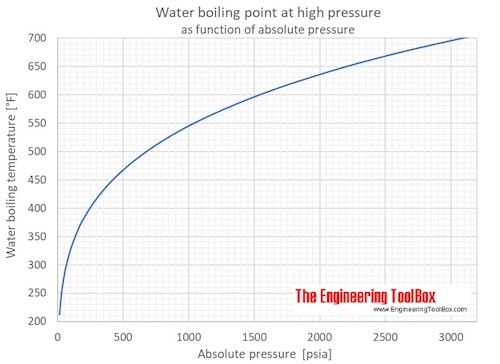
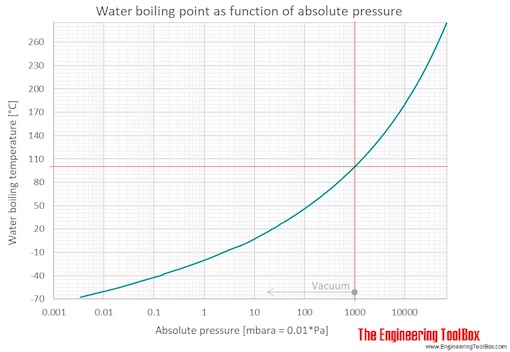
How does one calculate the boiling point of water at different pressures without a steam table? - Quora
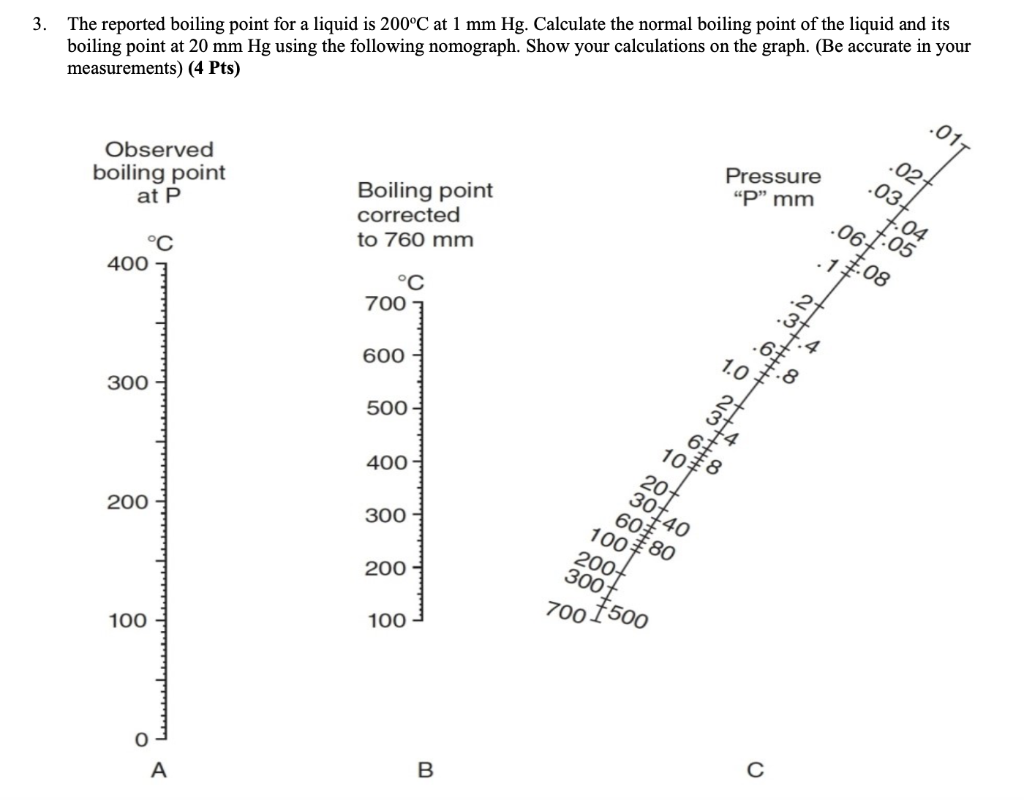
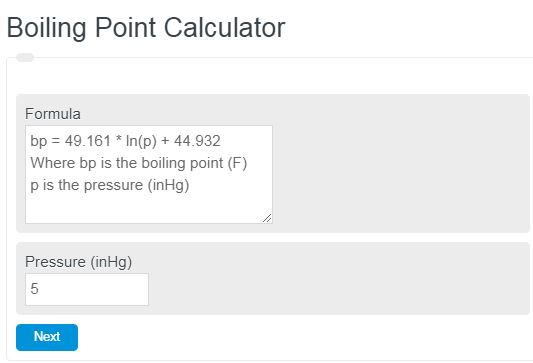
Problem Set #10 Assigned November 8, 2013 – Due Friday, November 15, 2013 Please show all work for credit To Hand in 1.