![PDF] Heats of combustion of liquid n-hexadecane, 1-hexadecene, n-decylbenzene, n-decylcyclohexane, n-decylcyclopentane, and the variation of heat of combustion with chain length | Semantic Scholar PDF] Heats of combustion of liquid n-hexadecane, 1-hexadecene, n-decylbenzene, n-decylcyclohexane, n-decylcyclopentane, and the variation of heat of combustion with chain length | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/50f5c61391a13518d9e65e9241e7cc44d2de1e09/2-Table1-1.png)
PDF] Heats of combustion of liquid n-hexadecane, 1-hexadecene, n-decylbenzene, n-decylcyclohexane, n-decylcyclopentane, and the variation of heat of combustion with chain length | Semantic Scholar

PDF) Relationship between heat of combustion, lignin content and burning weight loss | Jadwiga Fangrat - Academia.edu

The heats of combustion of polytetrafluoroethylene (teflon) and graphite in elemental fluorine - Journal of Research of NIST and Predecessor Publications - NIST Digital Archives

The heat of combustion of ethane gas at 373 k cal per mole. Assume that 50% of heat is lost, - YouTube

OneClass: The heat of combustion of octane, C_8H_18, can be measured in a way similar to the method y...

Heats of combustion and formation of liquid ethylenimine - Journal of Research of NIST and Predecessor Publications - NIST Digital Archives
Paper Heat Boiler Combustion Furnace Room, PNG, 1311x1365px, Paper, Area, Biomass, Boiler, Brand Download Free

Fire red hot orange burning flames heat core paper combustion furnace bonfire carbon charcoal black Stock Photo - Alamy
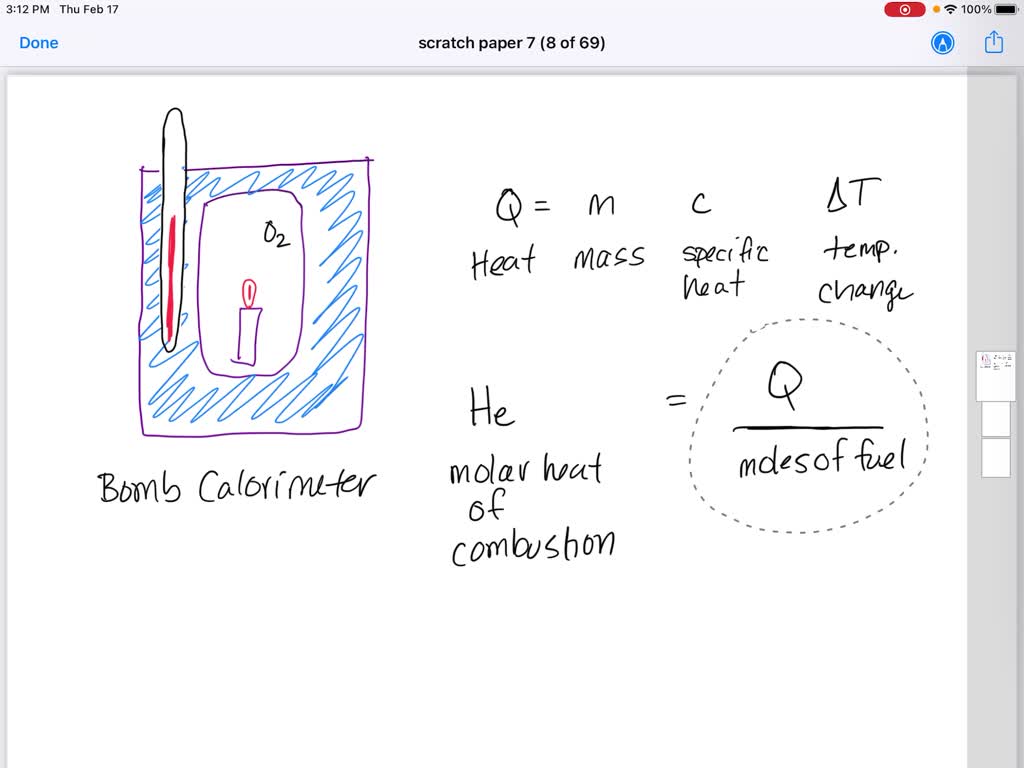
SOLVED: The quantity of heat released when a given amount of substance burns is called the heat of combustion and it is symbolized by ∆H comb. Paraffin, C 25 H 52, is
![PDF] Heats of combustion and formation of cellulose and nitrocellulose (cellulose nitrate) | Semantic Scholar PDF] Heats of combustion and formation of cellulose and nitrocellulose (cellulose nitrate) | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6d284ed89fb95d707df01192bd8c1a32e21b6e25/6-Table4-1.png)
PDF] Heats of combustion and formation of cellulose and nitrocellulose (cellulose nitrate) | Semantic Scholar

Heat of combustion of phosphorus and the enthalpies of formation of P4O10 and H3PO4 - Transactions of the Faraday Society (RSC Publishing)