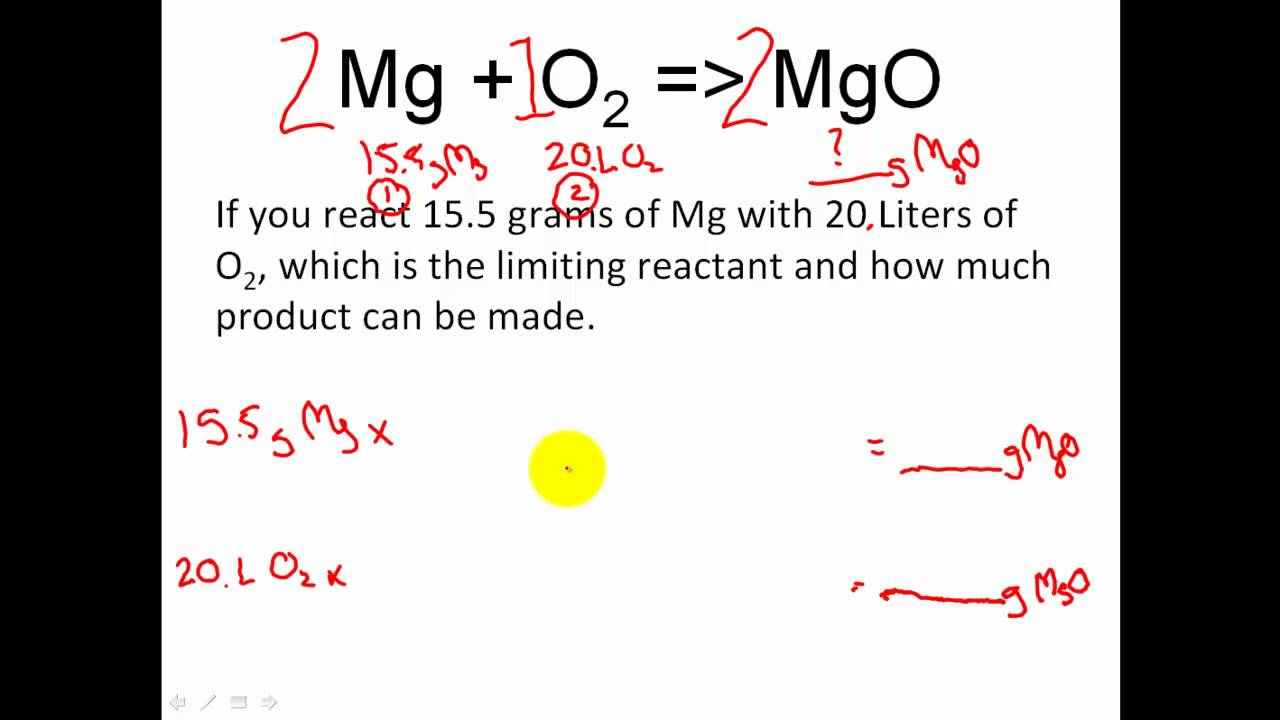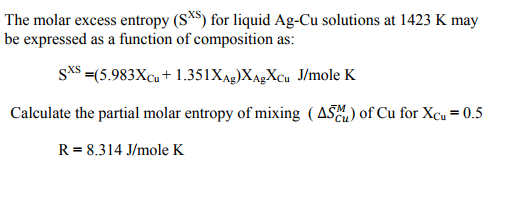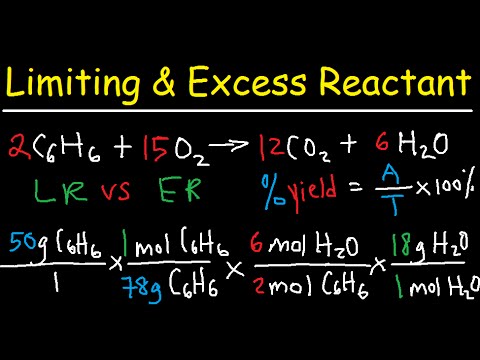6. 280 g of a mixture containing CA , and C_{2}H_{ 5:2 molar ratio is bumt in presence of excess ofoxygen. Calculate total moles of Co_{2} produced.(A)9 (B)T8 (C)7 (D) 12The | Snapsolve
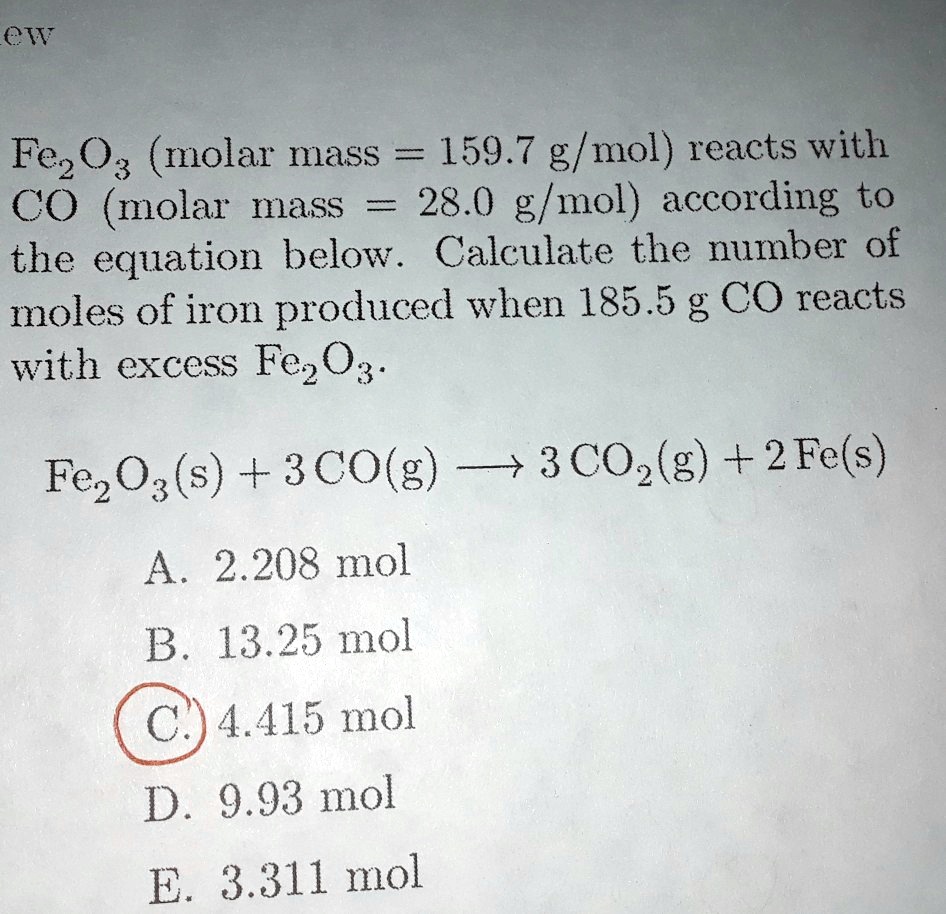
SOLVED: ew Fez O3 (molar mass = 159.7 g/mol) reacts with CO molar mass 28.0 g/mol) according to the equation below. Calculate the number of moles of iron produced when 185.5 g
R ational Interpolation F orm ulae for the Excess Functions o f B inary M ix tu res H. Brodowsky Institut für Physikalische Che
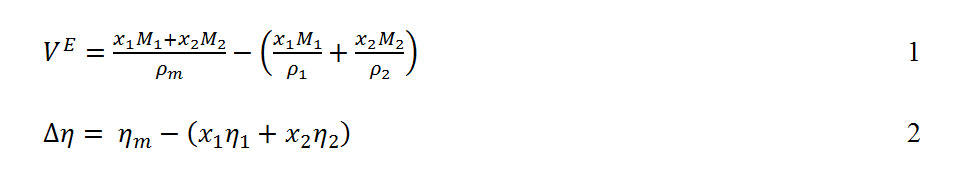
Derived thermodynamic properties of binary mixtures of m-Xylene, o-Xylene, and p-Xylene, with N,N-Dimethylformamide at T = (293.15, 303.15, 313.15 and 323.15) K : Oriental Journal of Chemistry

4. The excess molar Gibbs free energy of formation of solid solutions in the system Au-Cu can be represented by GX = -28280 XauXcu (J) Calculate the activities of Au and Cu
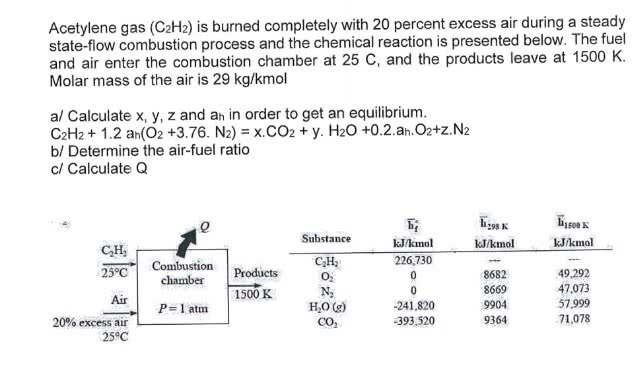
OneClass: Acetylene gas (C2H2) is burned completely with 20 percent excess air during a steady state-...