Effect of Tris, MOPS, and phosphate buffers on the hydrolysis of polyethylene terephthalate films by polyester hydrolases - Schmidt - 2016 - FEBS Open Bio - Wiley Online Library
Optimizing the culture environment in the IVF laboratory: impact of pH and buffer capacity on gamete and embryo quality
Buffer stability. (a) SenALiB was diluted in different buffers with... | Download Scientific Diagram

Figure 1 from (Un)suitability of the use of pH buffers in biological, biochemical and environmental studies and their interaction with metal ions – a review | Semantic Scholar
pH stability. a. Stability of FLIPO in different buffer systems in the... | Download Scientific Diagram

equilibrium - Why do buffers need to be composed of equal amounts of the acid and salt? - Chemistry Stack Exchange

![MOPS Buffer [3-(N-Morpholinopropanesulfonic Acid] - 1132-61-2 - Discovery Fine Chemicals MOPS Buffer [3-(N-Morpholinopropanesulfonic Acid] - 1132-61-2 - Discovery Fine Chemicals](https://discofinechem.com/wp-content/uploads/2015/08/1132-61-2.png)

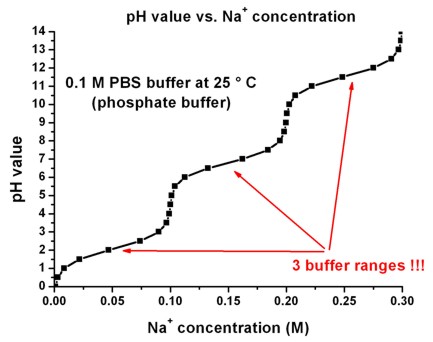


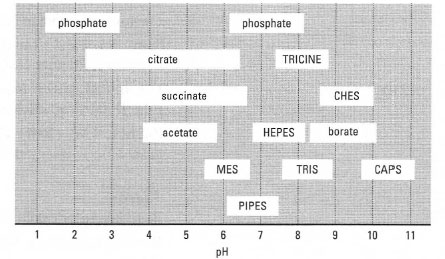


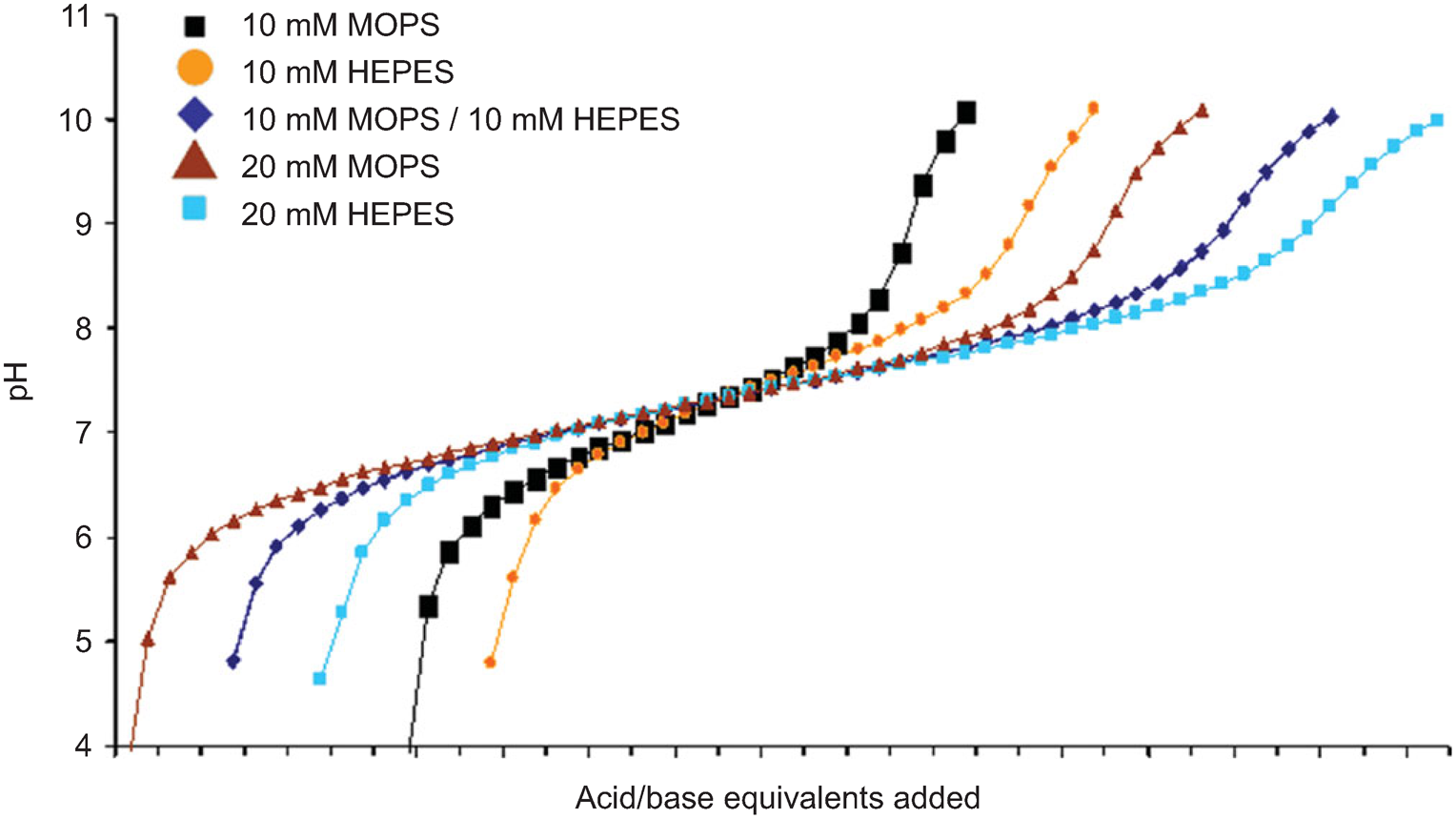


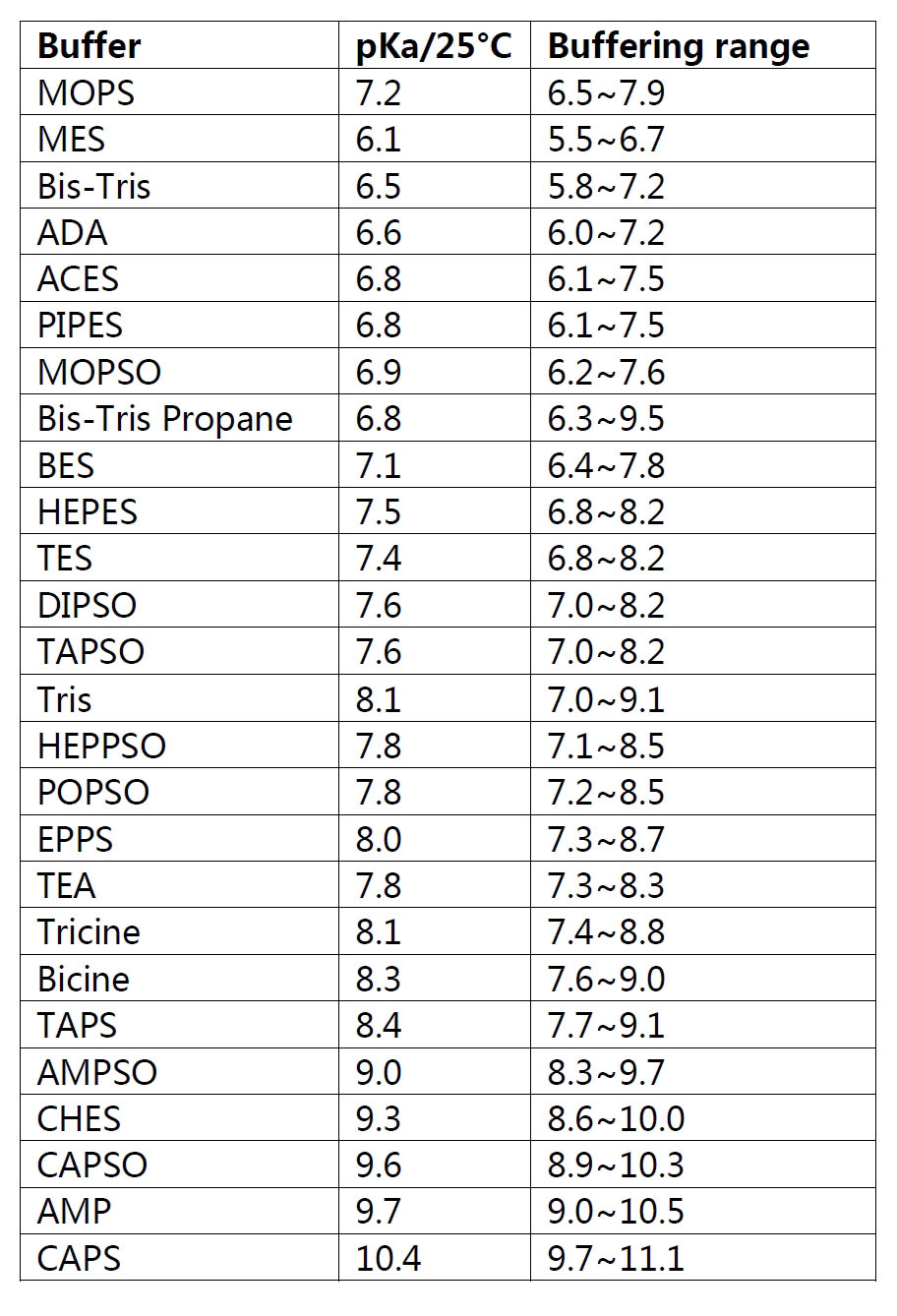
![Solved 7) MOPS (3-[N-morpholino]propanesulfonic) acid can be | Chegg.com Solved 7) MOPS (3-[N-morpholino]propanesulfonic) acid can be | Chegg.com](https://media.cheggcdn.com/study/3f1/3f1d2253-902e-4765-805c-d25932f0e9d3/image)