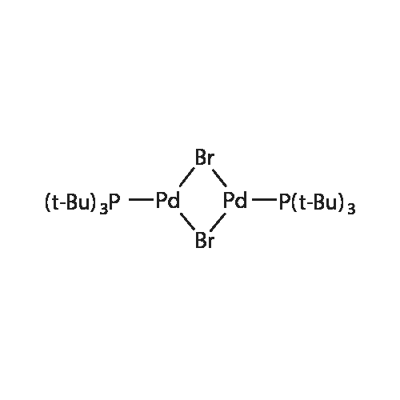
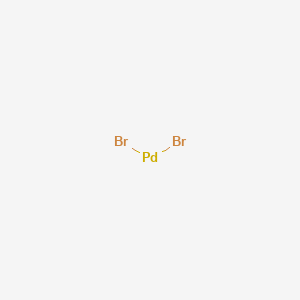3375-31-3 | Palladium(II) Acetate | Palladium(2+) Acetic Acid Salt; Palladium Acetic Acid Salt; Bis(Acetato)palladium; Diacetatopalladium; Diacetoxypalladium; HyCat 1; HyCat Base 1; Palladium Diacetate; Palladous Acetate; | (C₂H₃O₂)₂Pd | TRC

Palladium-Catalyzed Carbohalogenation: Bromide to Iodide Exchange and Domino Processes | Journal of the American Chemical Society
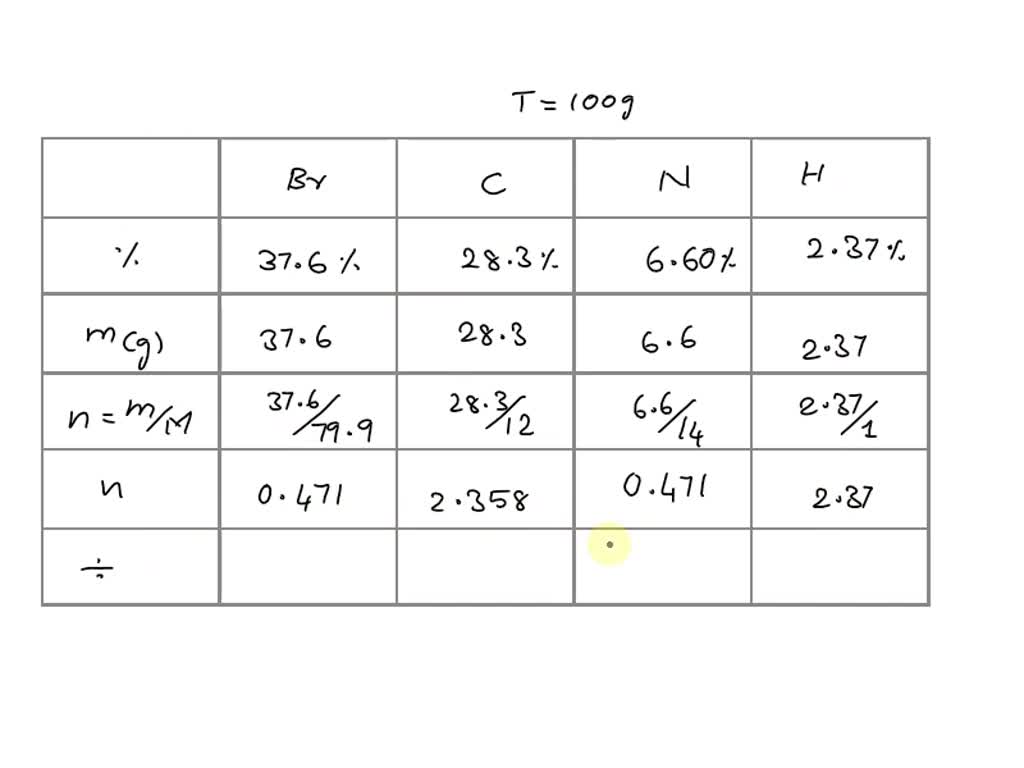
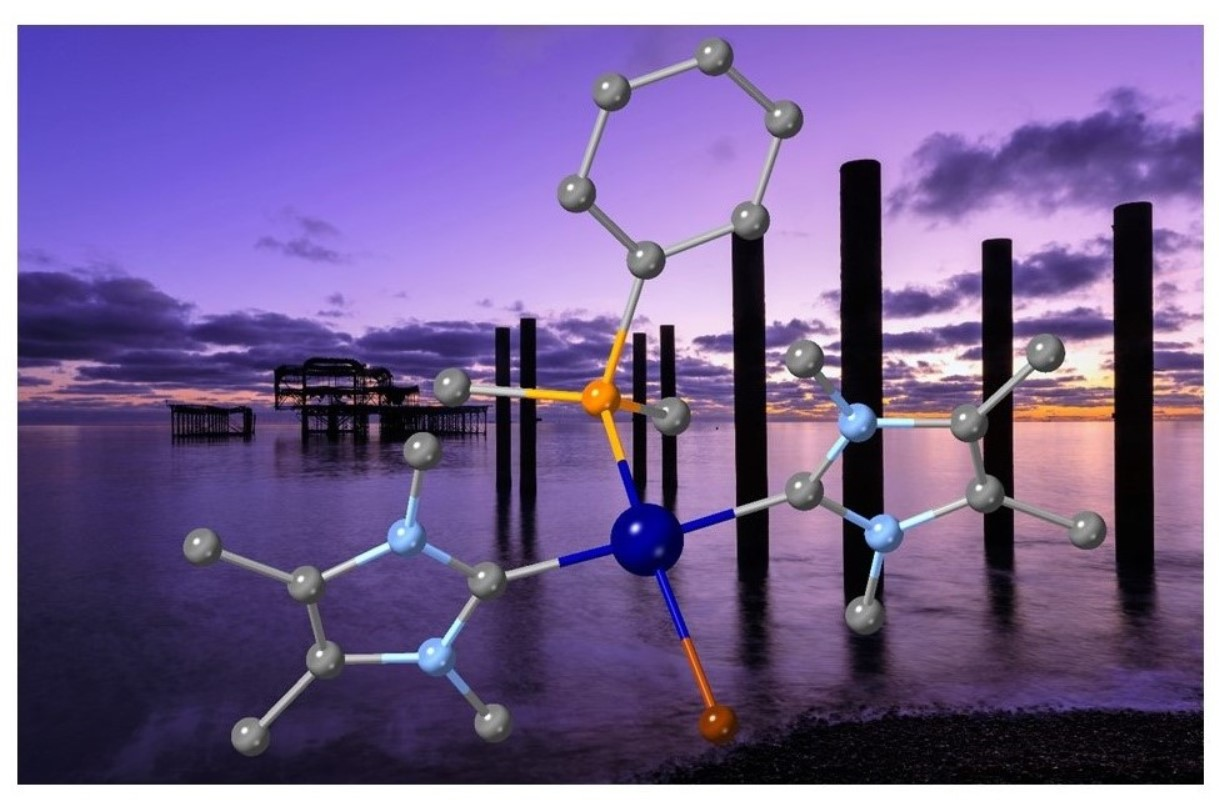
SOLVED: A palladium complex formed from a solution containing bromide ion and pyridine, C5H5N (a good electron-pair donor), is found on elemental analysis to contain 37.6% bromine, 28.3% carbon, 6.60% nitrogen, and
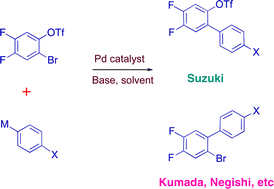
Aryl bromide/triflate selectivities reveal mechanistic divergence in palladium-catalysed couplings; the Suzuki–Miyaura anomaly - Chemical Communications (RSC Publishing)
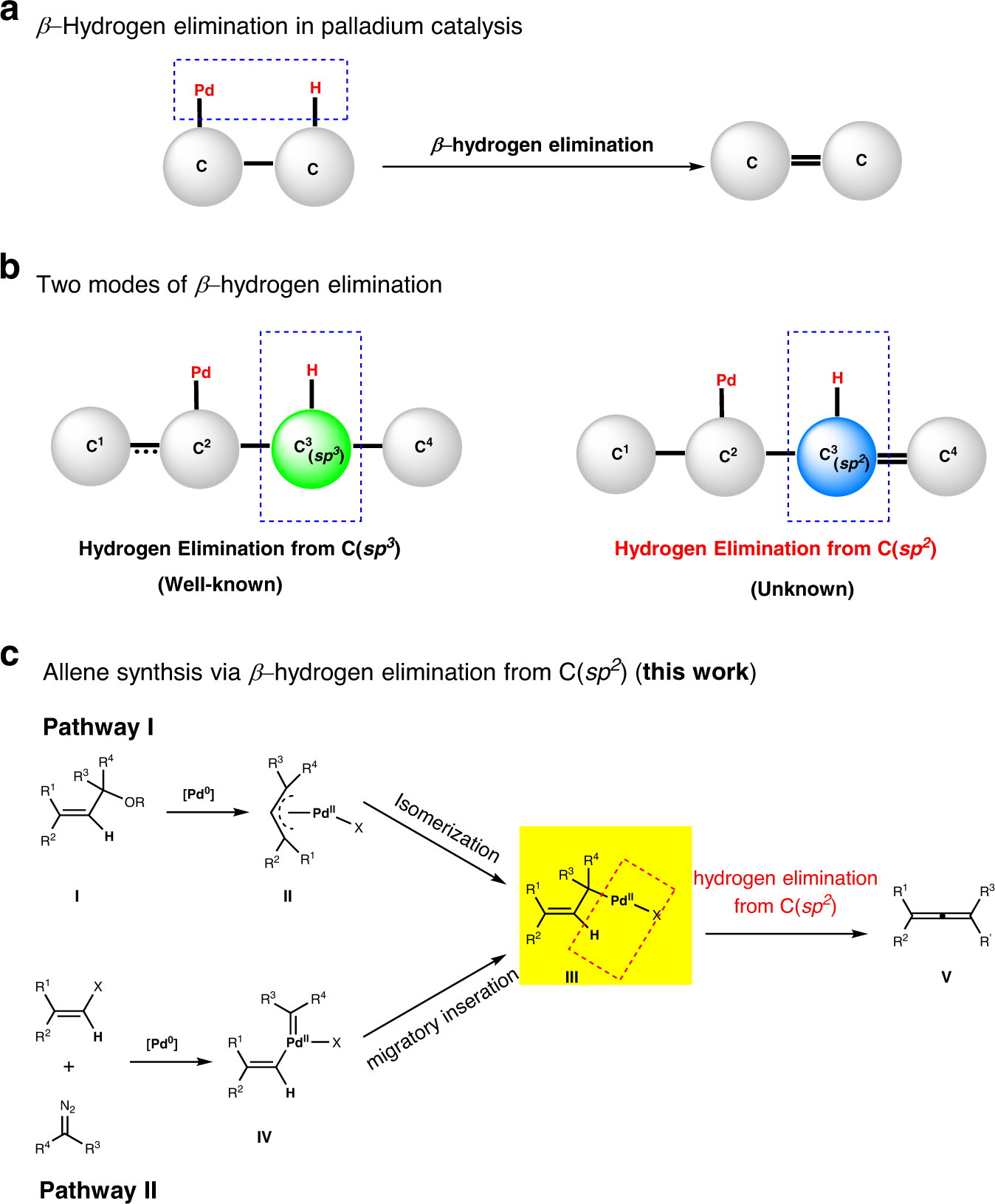
Palladium-catalyzed allene synthesis enabled by β-hydrogen elimination from sp2-carbon | Nature Communications

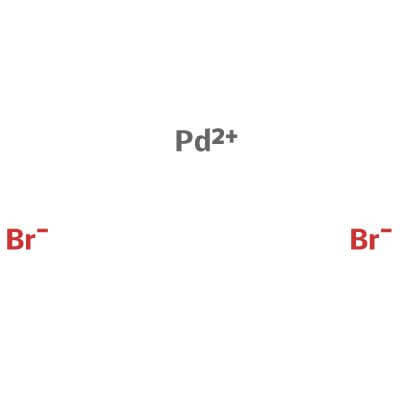
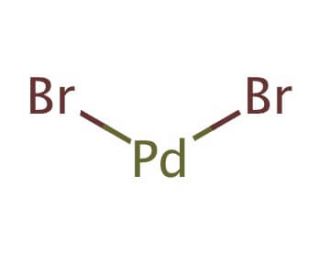
Palladium(II) bromide, Premion , 99.998% (metals basis), Pd 39.5% min, Thermo Scientific Chemicals, Quantity: 1 g | Fisher Scientific

Base-free efficient palladium catalyst of Heck reaction in molten tetrabutylammonium bromide - ScienceDirect