Representation of coordinated water molecules and polar interactions... | Download Scientific Diagram

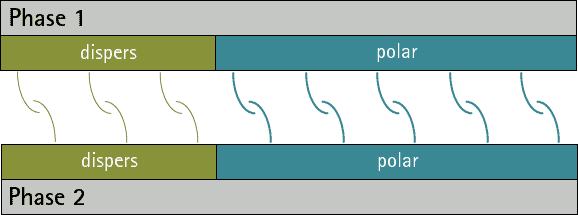
nanoHUB-U FoAFM L1.6: Tip-Surface Interactions (Non-Contact) - InterMolecular Interactions: Dispersion Force
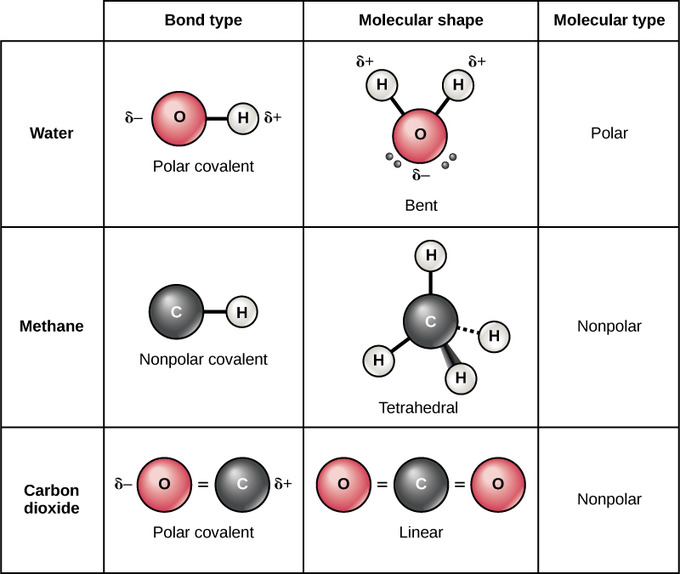
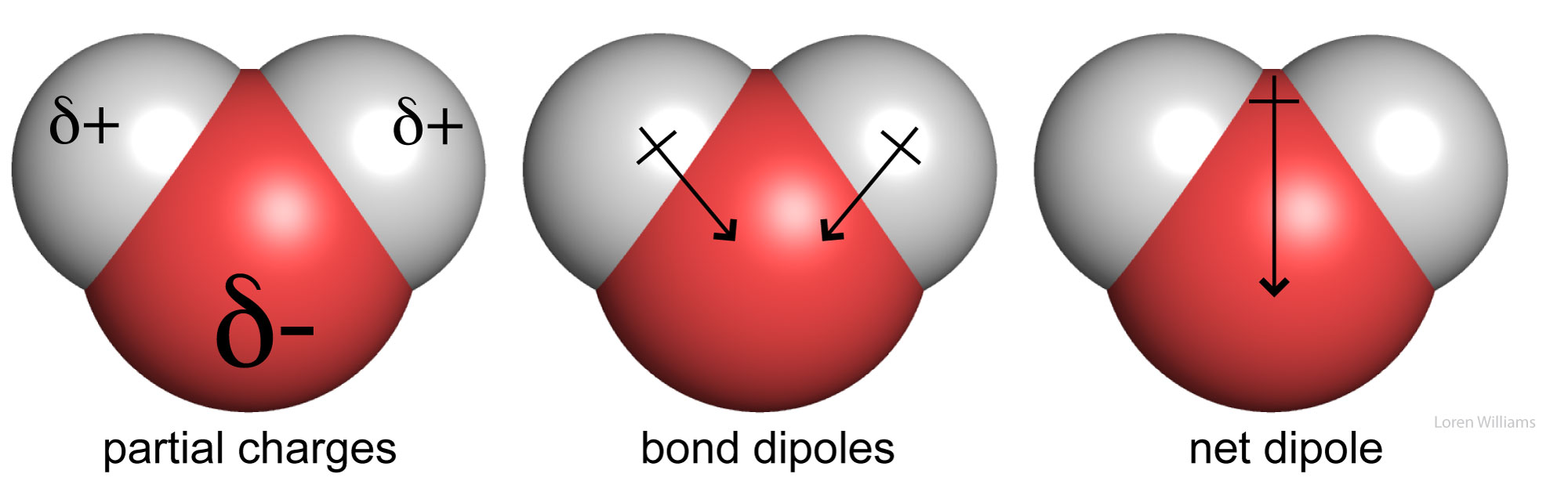
2.9: Atoms, Isotopes, Ions, and Molecules - Covalent Bonds and Other Bonds and Interactions - Biology LibreTexts

3D Interaction Homology: Hydropathic Analyses of the “π–Cation” and “π–π” Interaction Motifs in Phenylalanine, Tyrosine, and Tryptophan Residues | Journal of Chemical Information and Modeling

nanoHUB-U FoAFM L1.6: Tip-Surface Interactions (Non-Contact) - InterMolecular Interactions: Dispersion Force

SOLVED: Which of the: substances has polar interactions (dipole-dipole forces) between molecules? CHCI, I,O

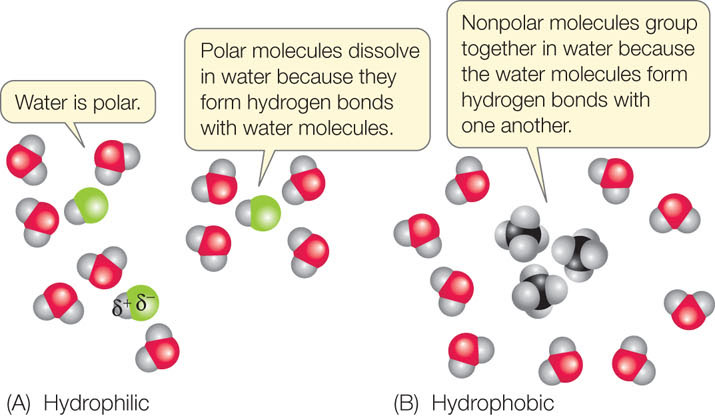
organic chemistry - If all intermolecular forces are electrostatic in nature, why don't large non polar molecule dissolve in water? - Chemistry Stack Exchange

The active site.: (A) The polar interactions between active sites and... | Download Scientific Diagram

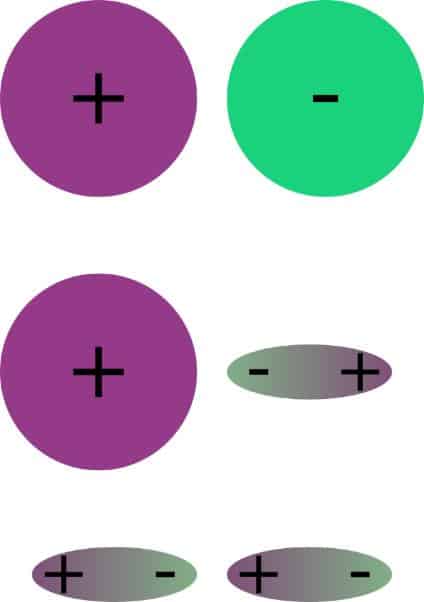
Van der Waals' Interactions Refers to all interactions between polar or nonpolar molecules, varying as r -6. Includes Keesom, Debye and dispersive interactions. - ppt download
On the Importance of Polar Interactions for Complexes Containing Intrinsically Disordered Proteins | PLOS Computational Biology
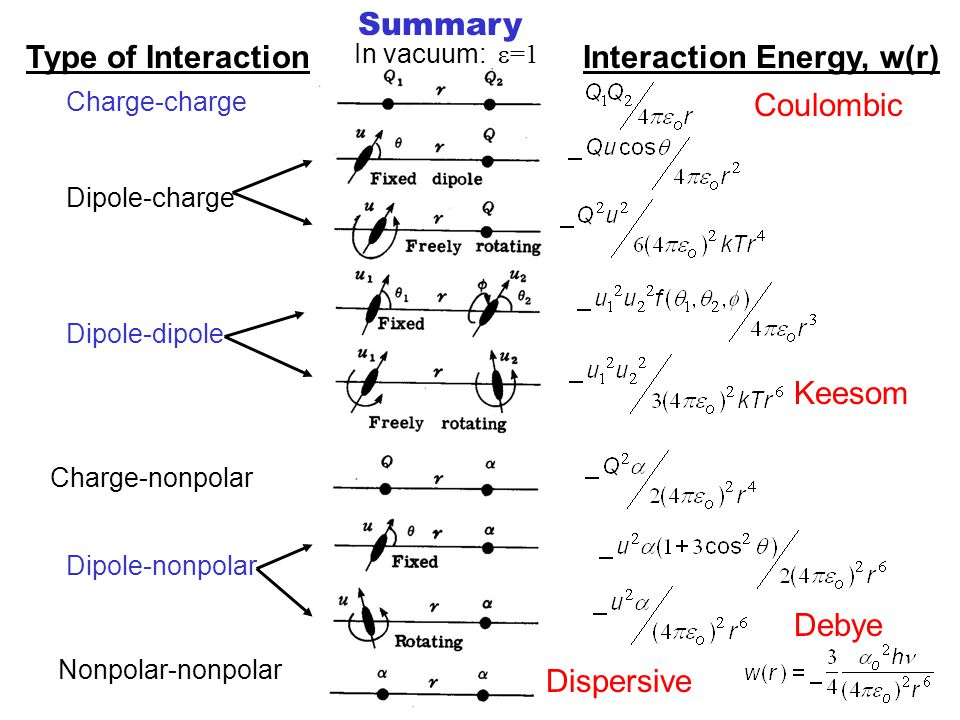
Van der Waals' Interactions Refers to all interactions between polar or nonpolar molecules, varying as r -6. Includes Keesom, Debye and dispersive interactions. - ppt download