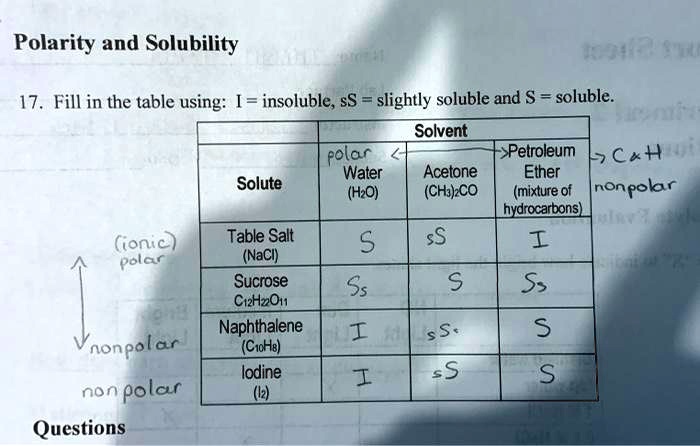
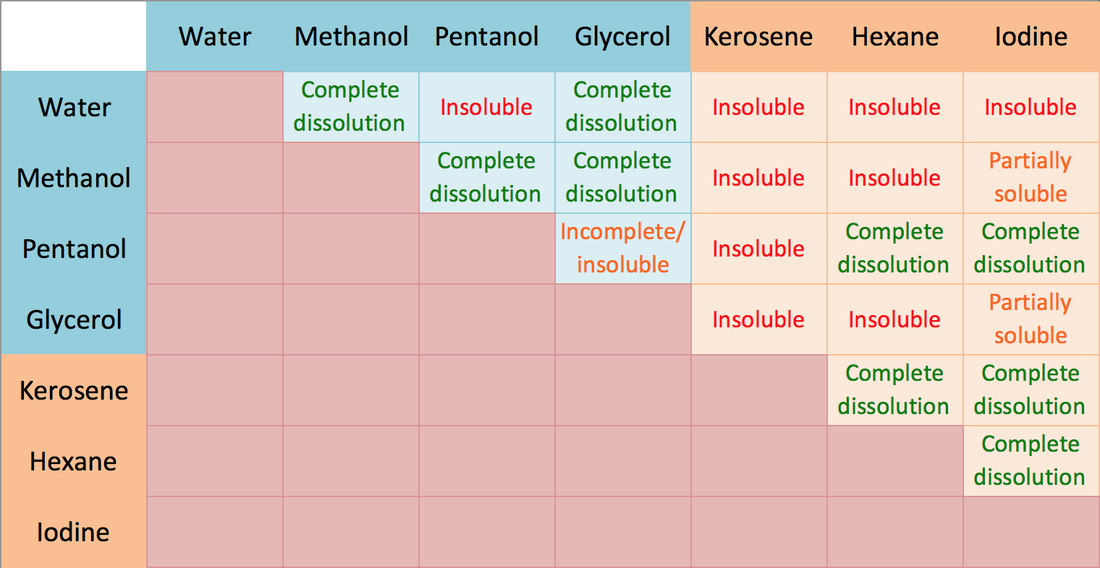
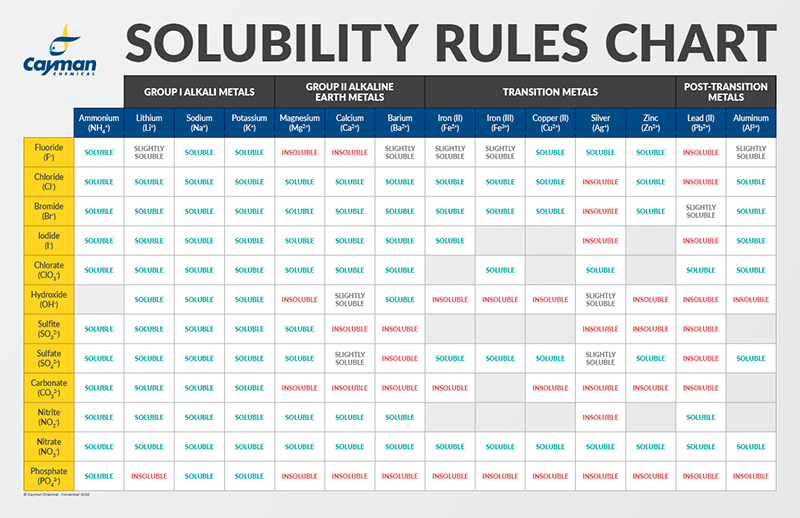
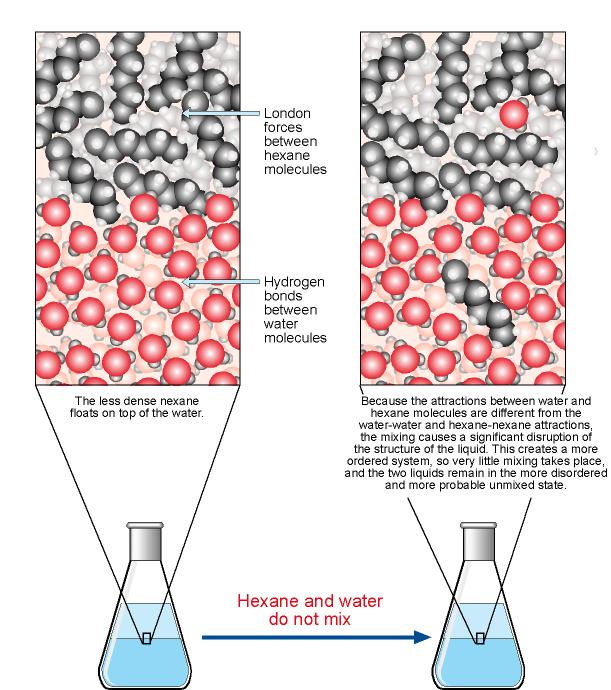
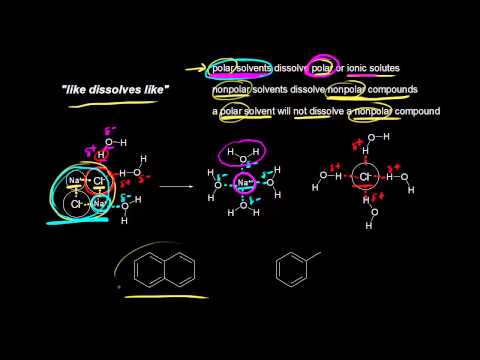
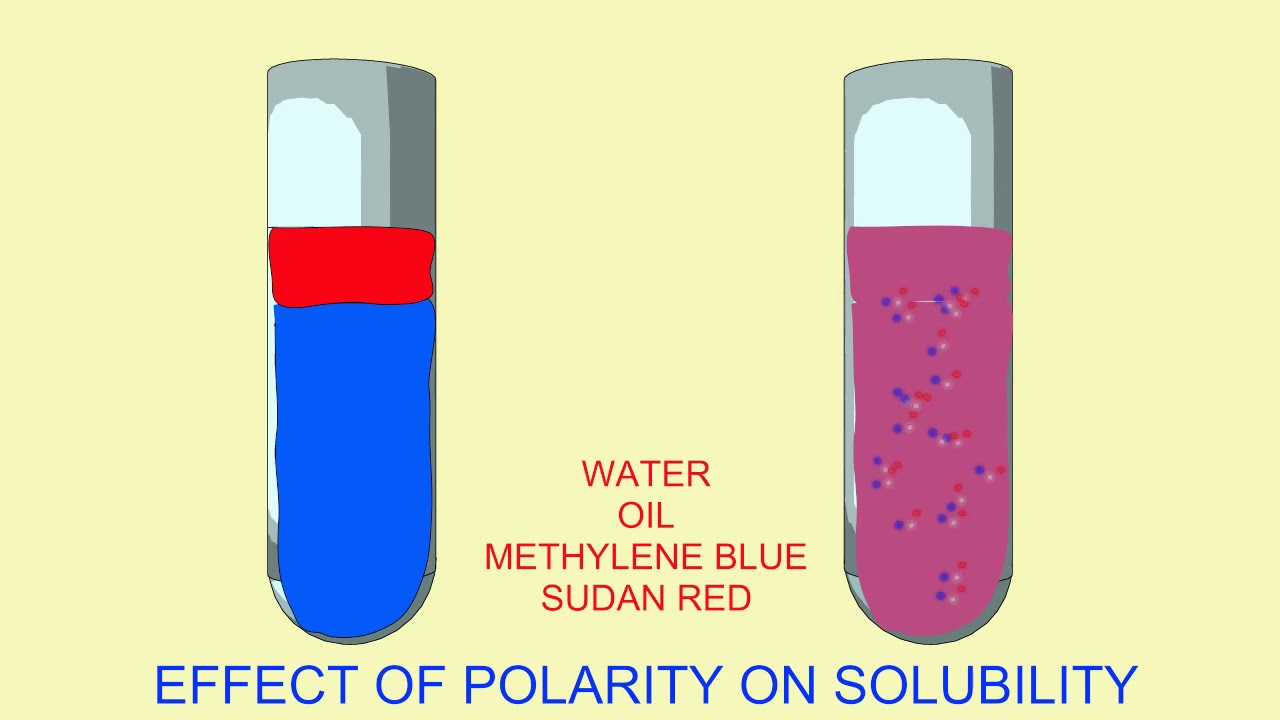
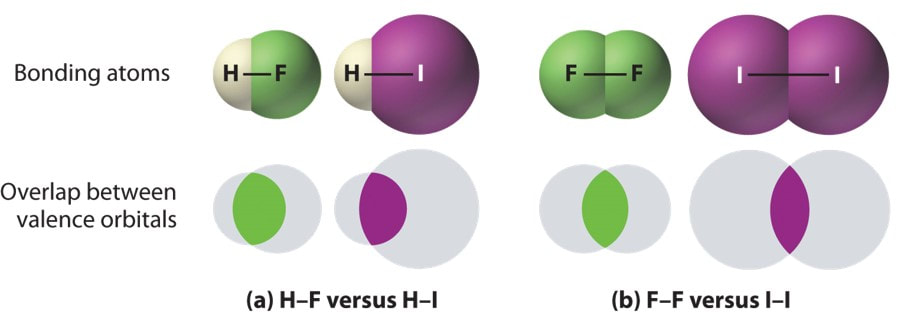
Polarity of water Solubility.PNG - Polarity of Water: Solubility The polar nature of water allows it to dissolve polar substances . Water does not | Course Hero

The relation between solubility, polarity, and molar mass for various... | Download Scientific Diagram

Impact of Dynamically Exposed Polarity on Permeability and Solubility of Chameleonic Drugs Beyond the Rule of 5 | Journal of Medicinal Chemistry

SOLVED: Table 2. Solubility and polarity of compound Solubility in Polar or non-polar cycloherane solute Solubillty In water Choose Choose. KMno4 Soluble Insoluble choose Choose: Insoluble Soluble Choo1e Choose: Sucrose Soluble Insoluble

Solubility of Polar and Nonpolar Aromatic Molecules in Subcritical Water: The Role of the Dielectric Constant | Journal of Chemical Theory and Computation