Peripheral Neuropathy Treatment Market Growth Set to Surge Significantly US$ 2,425.9 Million by 2030, Future Trends, Key Insights and Business Strategies for Teva Pharmaceutical Industries Ltd., Eli Lilly and Company, Johnson & Johnson
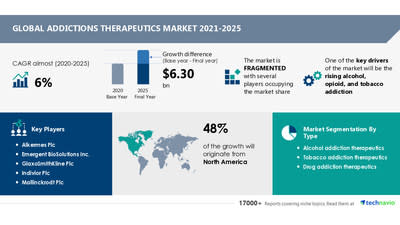
Addictions Therapeutics Market: 48% of Growth to Originate from North America | COVID-19 Impact and Global Analysis by Type and Geography
More Options For Opioid Overdose: Nasal Naloxone On Brink Of OTC Use, While Opiant And Orexo Stay Rx To Counter High-Potency Opioids :: Pink Sheet

Bioprojet SCR:Company Profile & Technical Research,Competitor Monitor,Market Trends - Discovery | PatSnap

PDF) Long-Term Safety and Efficacy of Subcutaneous Methylnaltrexone in Patients with Opioid-Induced Constipation and Chronic Noncancer Pain: A Phase 3, Open-Label Trial