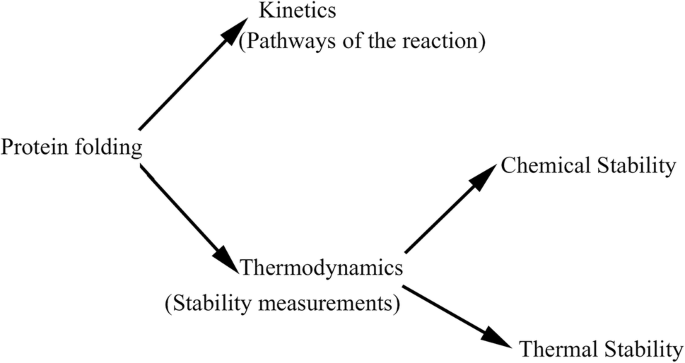
Thermodynamics of protein folding: methodology, data analysis and interpretation of data | SpringerLink
![PDF] Effect of glycosylation on protein folding: A close look at thermodynamic stabilization | Semantic Scholar PDF] Effect of glycosylation on protein folding: A close look at thermodynamic stabilization | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/5077610fcb706a77fe53db3511d3c260a744f898/2-Figure1-1.png)
PDF] Effect of glycosylation on protein folding: A close look at thermodynamic stabilization | Semantic Scholar
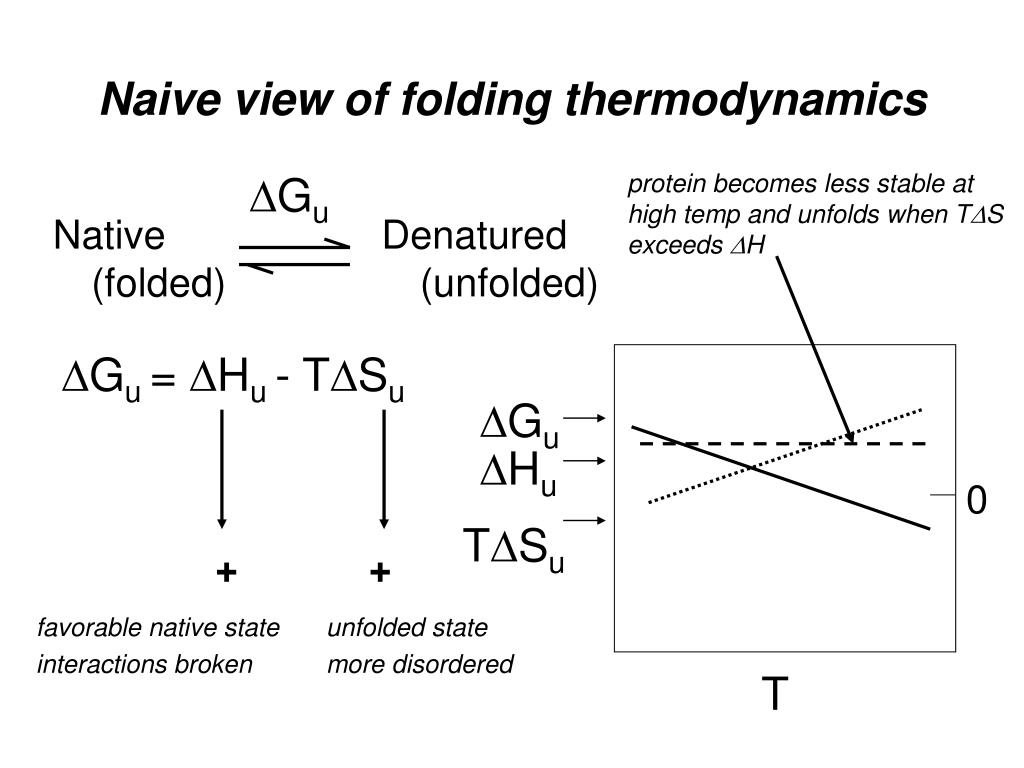
PPT - Basic protein structure and stability VI: Thermodynamics of protein stability PowerPoint Presentation - ID:247919

Size-dependent macromolecular crowding effect on the thermodynamics of protein unfolding revealed at the single molecular level - ScienceDirect

PPT – Protein folding: Review of thermodynamics PowerPoint presentation | free to view - id: 1d1384-ZDc1Z

Small Neutral Crowding Solute Effects on Protein Folding Thermodynamic Stability and Kinetics | The Journal of Physical Chemistry B
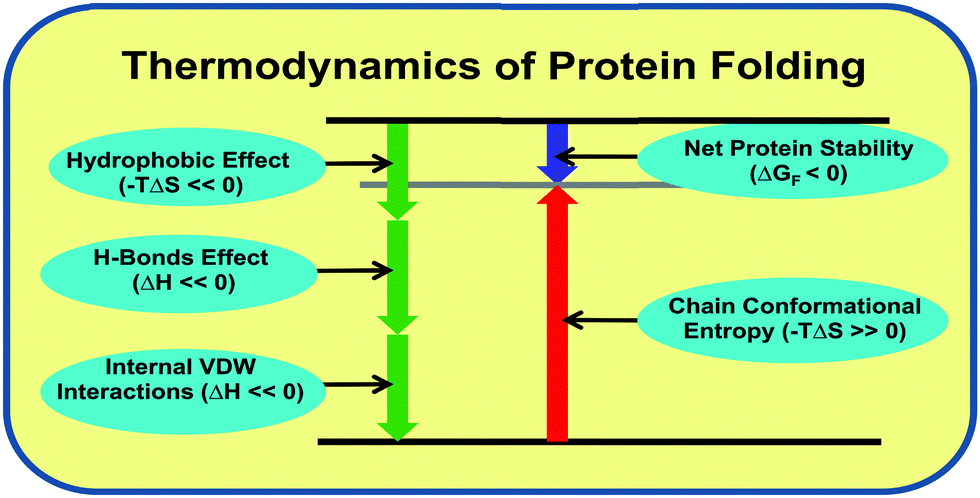
Changing relations between proteins and osmolytes: a choice of nature - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C8CP02949K
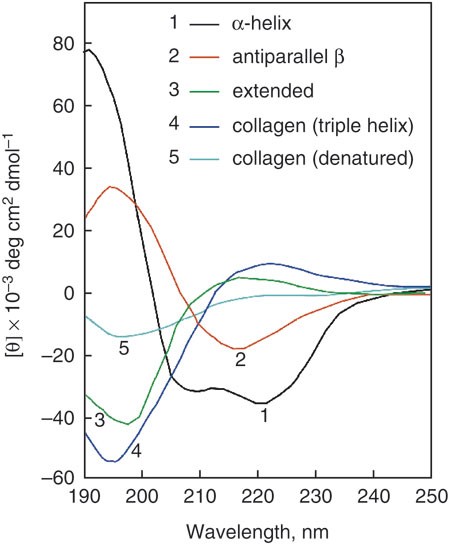
Using circular dichroism collected as a function of temperature to determine the thermodynamics of protein unfolding and binding interactions | Nature Protocols



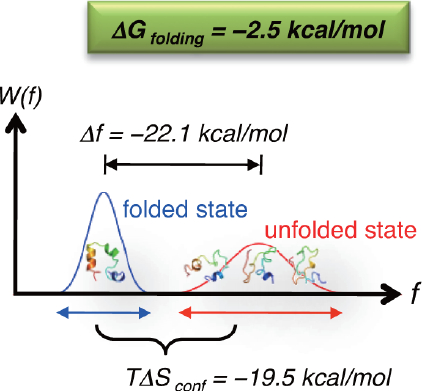



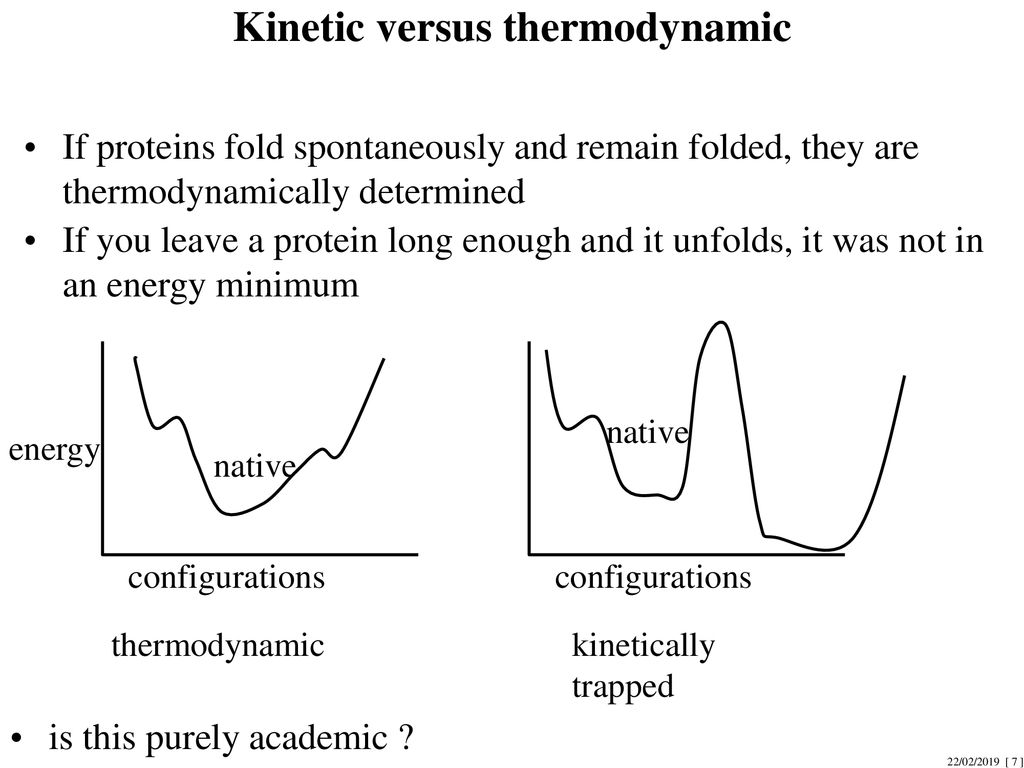
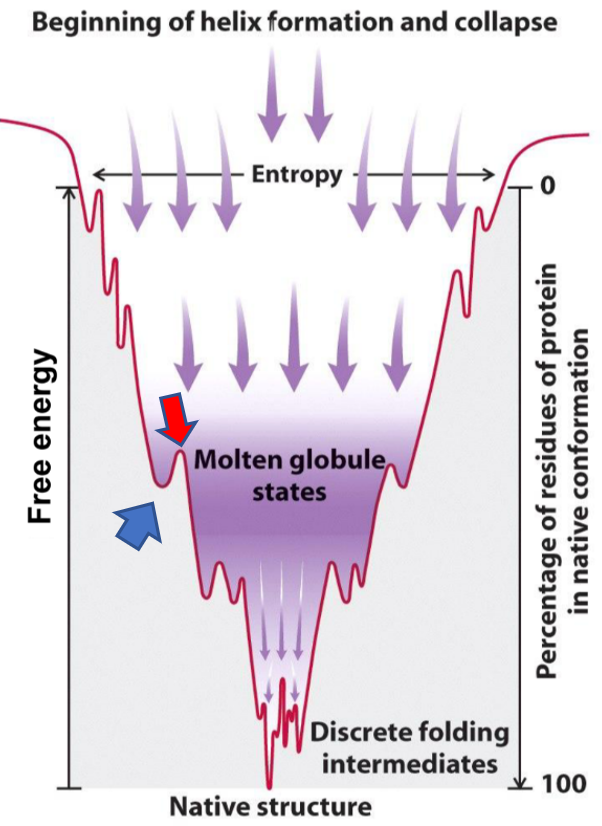
![PDF] Kinetics versus thermodynamics in protein folding. | Semantic Scholar PDF] Kinetics versus thermodynamics in protein folding. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7f06324d972547a8a75ac5ca00d7a05d201e4744/3-Figure2-1.png)
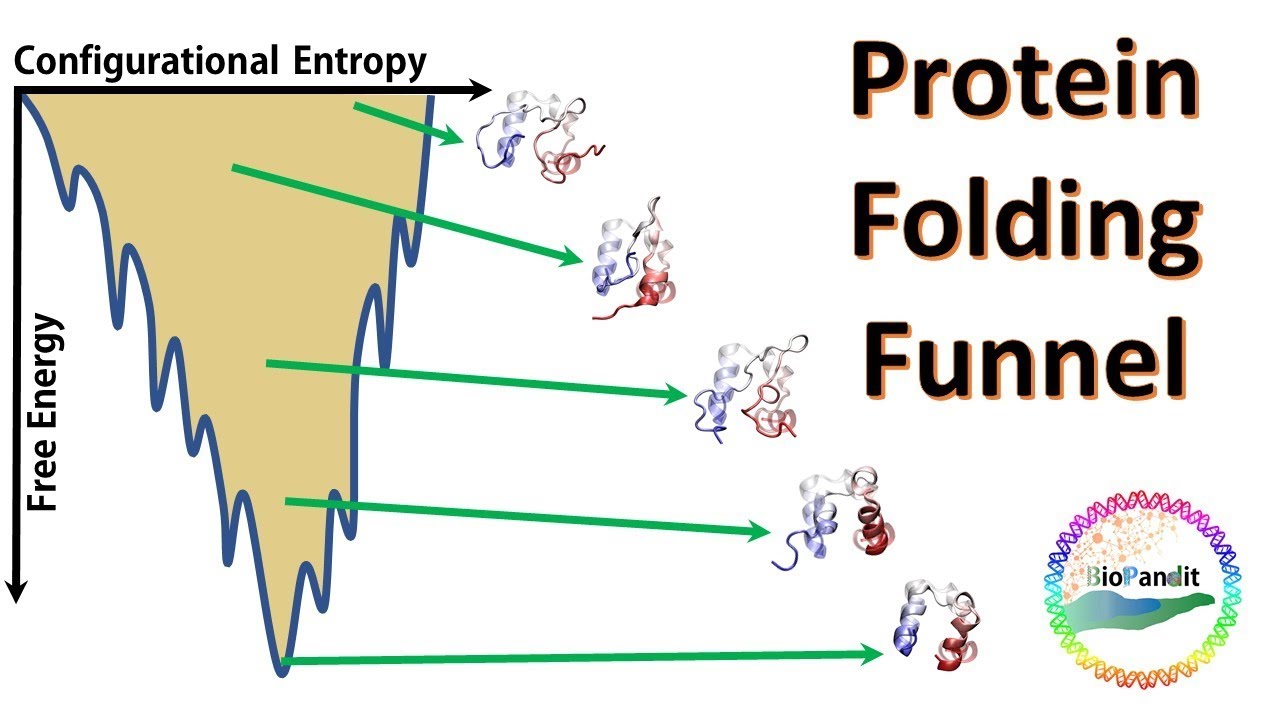
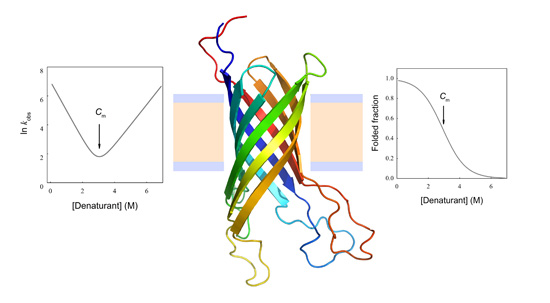
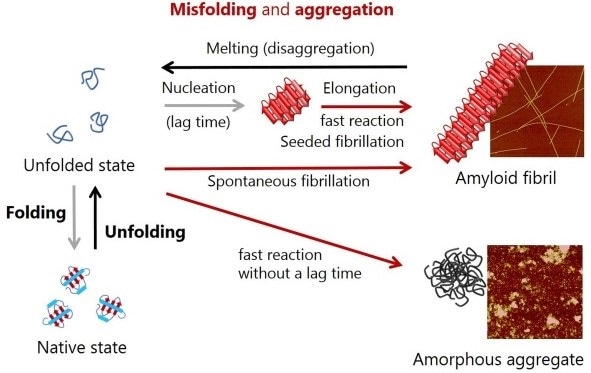
![PDF] Kinetics versus thermodynamics in protein folding. | Semantic Scholar PDF] Kinetics versus thermodynamics in protein folding. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/7f06324d972547a8a75ac5ca00d7a05d201e4744/2-Figure1-1.png)