Xenon Tetroxide Peroxymonosulfuric Acid Glycerol Oxyacid PNG, Clipart, Acid, Active Ingredient, Angle, Area, Black And White
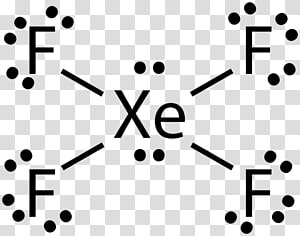
Chemistry Cartoon png download - 1000*1000 - Free Transparent Xenon Hexafluoride png Download. - CleanPNG / KissPNG

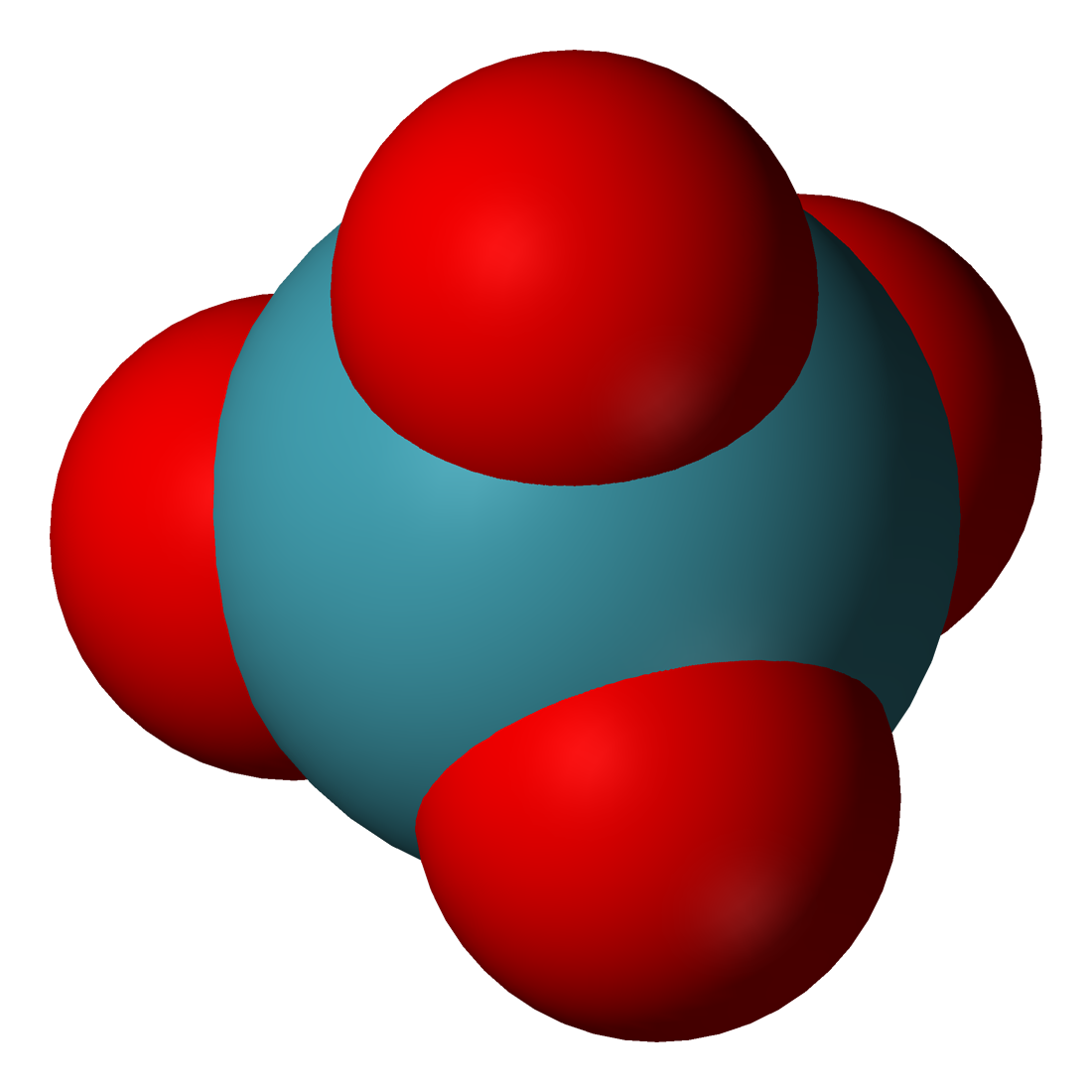
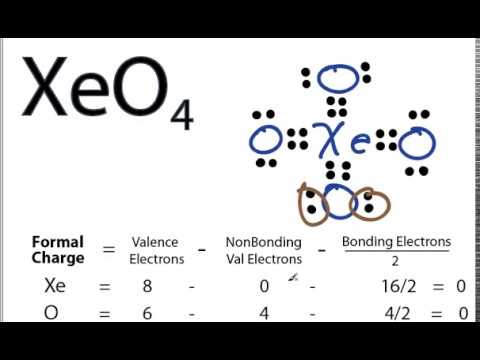
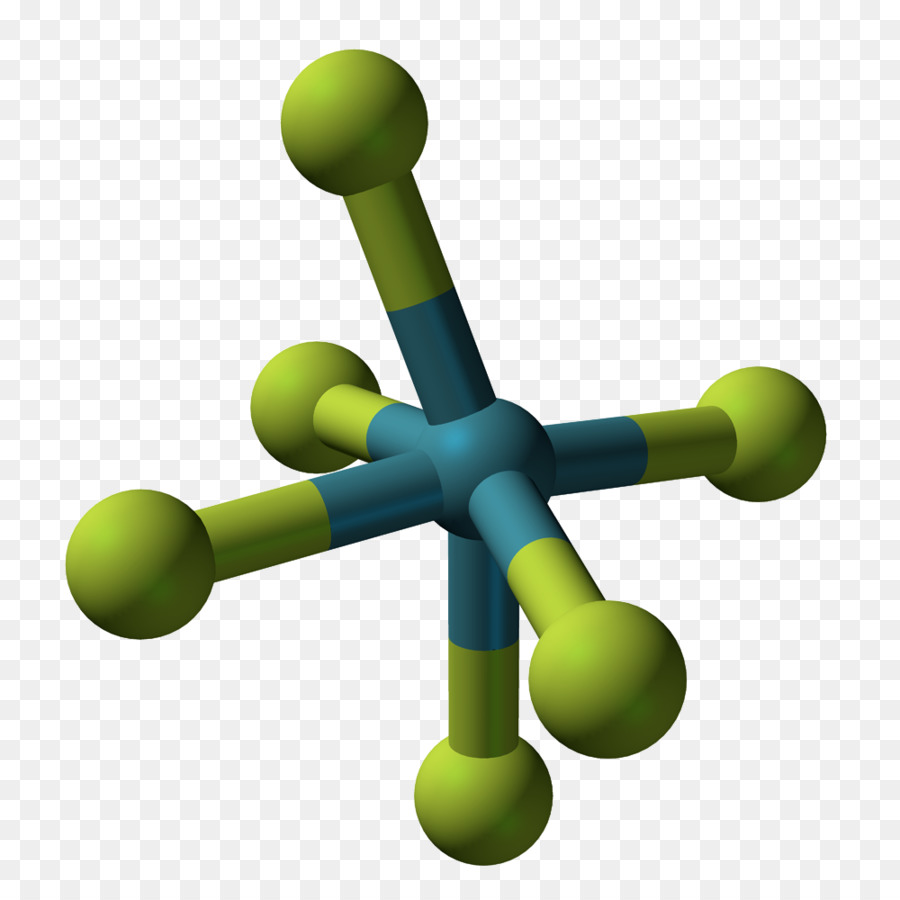
Draw the Lewis structure, predict the molecular structure, and describe the bonding (in terms of the hybrid orbitals for the central atom) for XeO4. | Homework.Study.com
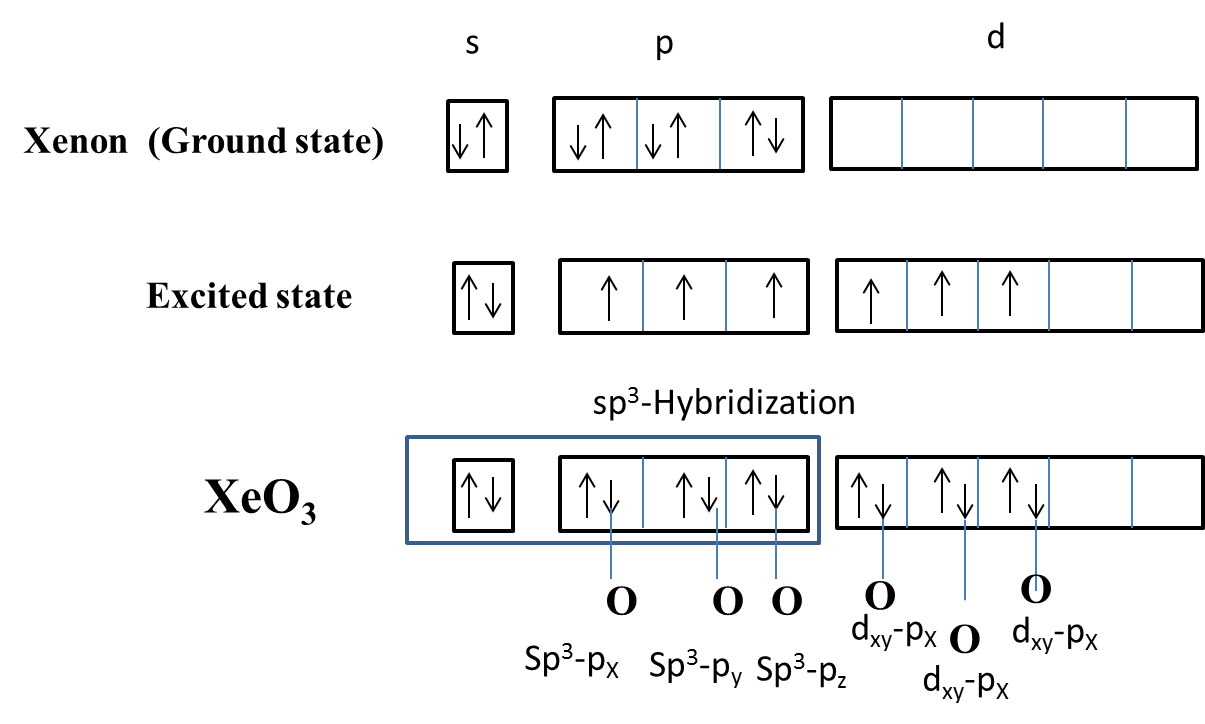
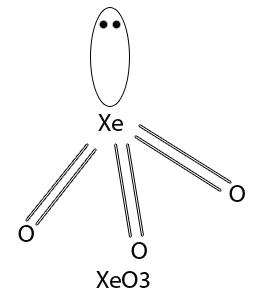
Which of the following overlaps is not present in $Xe{{O}_{3}}$ molecule?A. $s{{p}^{3}}+{{p}_{x}}$ B. $s{{p}^{3}}+{{p}_{y}}$C. ${{d}_{xz}}+{{p}_{x}}$D. $s{{p}^{3}}+s$

Xenon hexafluoride Sulfur hexafluoride Xenon tetrafluoride, vi, hexafluoride, xenon Hexafluoride, xenon png | PNGWing
Xenon Tetroxide Molecule Chemical Compound Chemistry, PNG, 1100x701px, Xenon Tetroxide, Area, Black And White, Brand, Chemical

SOLVED: Nrite a formula for each molecular compound (a) chlorine monoxide (b) xenon tetroxide (c) xenon hexafluoride (d) carbon tetrabromide (e) diboron tetrachloride (f) tetraphosphorus triselenide

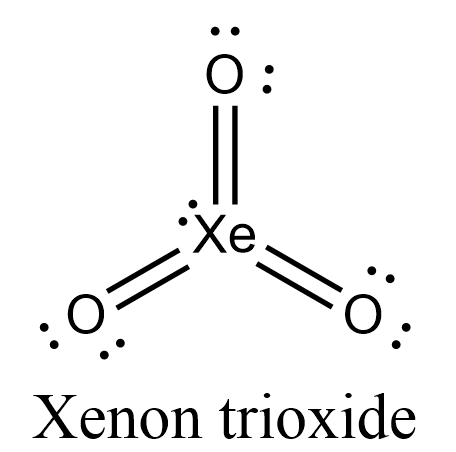
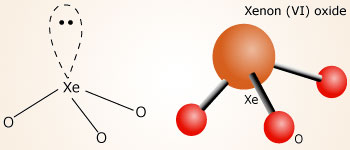
Draw four possible Lewis structures of the molecule xenon trioxide, XeO3, one each with zero, one, two, or - Brainly.com
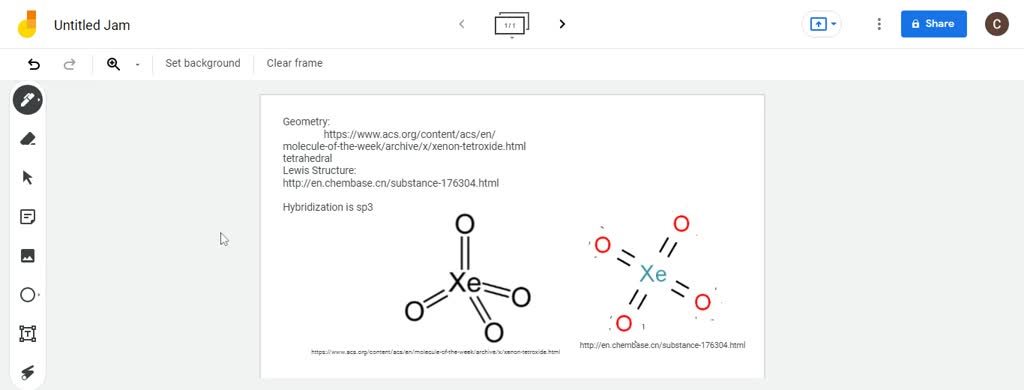
SOLVED:Xenon tetroxide, XeO4, is a colorless, unstable gas. Give the Lewis formula for the XeO4 molecule. What is the hybridization of the xenon atom in this compound? What geometry would you expect